Identification of a BAZ2A-Bromodomain Hit Compound by Fragment Growing.
Dalle Vedove, A., Cazzanelli, G., Batiste, L., Marchand, J.R., Spiliotopoulos, D., Corsi, J., D'Agostino, V.G., Caflisch, A., Lolli, G.(2022) ACS Med Chem Lett 13: 1434-1443
- PubMed: 36105334
- DOI: https://doi.org/10.1021/acsmedchemlett.2c00173
- Primary Citation of Related Structures:
7B7B, 7B7G, 7B7I, 7B82, 7BC2, 7QVT, 7QVU, 7QVV, 7QWF, 7QWU, 7QWY, 7QX2, 7QX9, 7QXL, 7QYE, 7QYO, 7QYT, 7QYU, 7QYV, 7QYW, 7QZ0, 7QZ4, 7QZB, 7QZC, 7QZI, 7QZT, 7R01, 7R0B - PubMed Abstract:
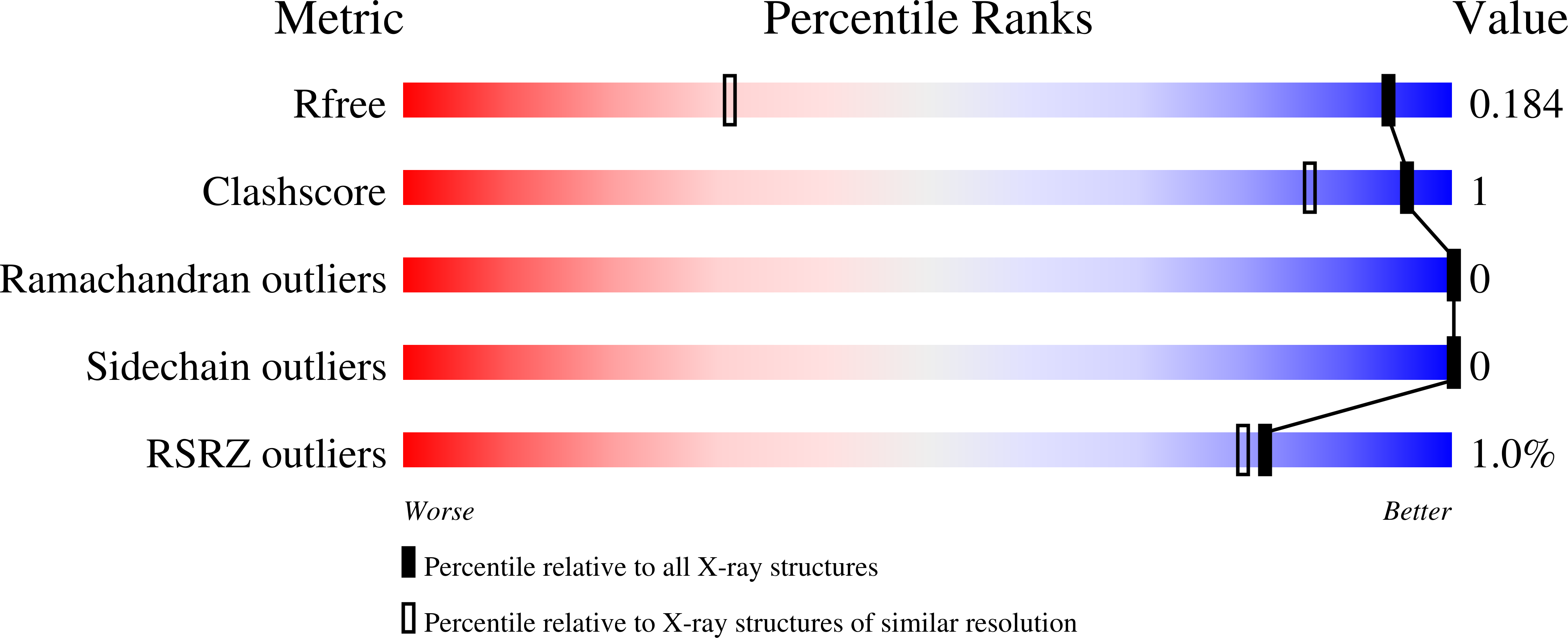
BAZ2A is an epigenetic regulator affecting transcription of ribosomal RNA. It is overexpressed in aggressive and recurrent prostate cancer, promoting cellular migration. Its bromodomain is characterized by a shallow and difficult-to-drug pocket. Here, we describe a structure-based fragment-growing campaign for the identification of ligands of the BAZ2A bromodomain. By combining docking, competition binding assays, and protein crystallography, we have extensively explored the interactions of the ligands with the rim of the binding pocket, and in particular ionic interactions with the side chain of Glu1820, which is unique to BAZ2A. We present 23 high-resolution crystal structures of the holo BAZ2A bromodomain and analyze common bromodomain/ligand motifs and favorable intraligand interactions. Binding of some of the compounds is enantiospecific, with affinity in the low micromolar range. The most potent ligand has an equilibrium dissociation constant of 7 μM and a good selectivity over the paralog BAZ2B bromodomain.
Organizational Affiliation:
Department of Cellular, Computational and Integrative Biology - CIBIO, University of Trento, via Sommarive 9, 38123 Povo - Trento, Italy.